/ MEDICAL DEVICE COMPANY / ACQUIRED BY TELEFLEX (NASDAQ: TFX) IN 2022
May 17, 2023
Cincinnati-based bariatric surgeon Jonathan Thompson, M.D., founded Standard Bariatrics in 2014 to address key unmet needs for weight-loss surgery confirmed by his bariatric surgeon colleagues. He attracted seed investors from the local biotech community to fund the company, hire key employees, and develop a first-generation device. The greater challenge was getting the company’s lead product Titan SGS® developed and financed all the way through commercialization. Here’s how that happened and the role RiverVest played.
THE SNAPSHOT
- Founded in 2014 by Cincinnati surgeon Jon Thompson, M.D.
- $20 million Series A led by RiverVest in 2018
- $35 million Series B in 2021
- Acquired by Teleflex for $170 million in 2022 plus additional milestone payments
THE IMPACT
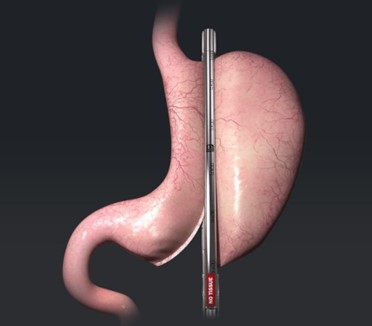
Standard Bariatrics’ medical devices are designed specifically for gastric tissue. Bariatric surgeons realize that consistent, repeatable results and more consistent and symmetrical sleeve pouch anatomy deliver better outcomes for their patients undergoing gastric sleeve weight-loss surgery and for the clinics where these procedures are performed.