/ BIOPHARMA COMPANY / ACQUIRED BY ROCHE (SIX: RO, ROG; OTCQX: RHHBY) IN 2022
October 6, 2023
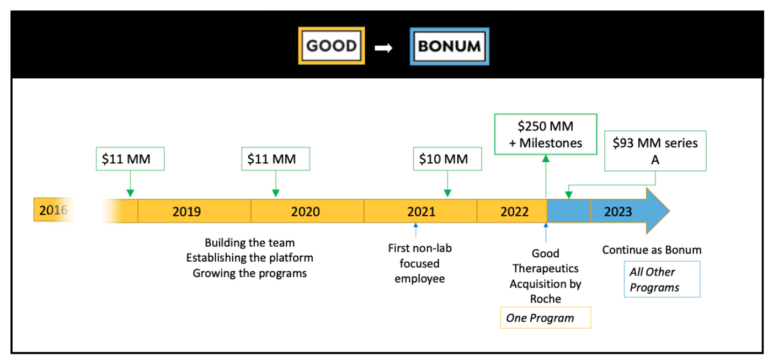
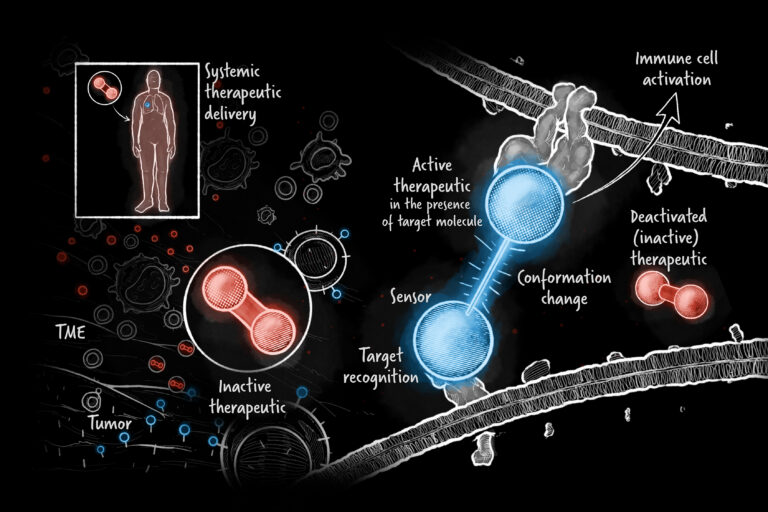
Good Therapeutics, founded in 2016 by molecular biologist John Mulligan, Ph.D., was acquired by Roche in 2022 for $250 million upfront plus potential milestone payments. Even better, Mulligan and company retained their platform and the ability to mine it for innovative drugs to fight cancer and other diseases. The potential for conditionally active protein technology is vast and could be game-changing for patients. Here’s a peek at how Good’s acquisition played out and what the future may hold for its spinout, Bonum Therapeutics.
INVESTMENT SNAPSHOT
- $600,000 in Seed funding
- $22 million in Series A financing, co-led by RiverVest, and $10 million Series B round
- $250 million (8x upfront) acquisition by Roche plus potential milestone payments
- $93 million Series A financing for spinout company, Bonum, with original syndicate plus one new investor participating
- Investment Syndicate: Codon Capital, Digitalis Venture, RiverVest Venture Partners, Roche Venture Fund, and 3x5 Partners